What is hydrogen
Hydrogen (H₂) is the most abundant element in the universe, accounting for approximately 75% of all matter by mass. On Earth, hydrogen does not exist in its pure elemental form but is instead found in compounds such as water (H₂O), hydrocarbons (C-H), and various organic substances.
Hydrogen gas is a colorless, odorless, tasteless, non-toxic substance and the lightest of all chemical elements.

Key Properties of Hydrogen Gas (H₂)
- Hydrogen is the lightest known gas, approximately 14 times lighter than air.
- Hydrogen burns vigorously in oxygen, producing only water (H₂O) and a significant amount of energy.
- Hydrogen acts as a powerful reducing agent, capable of reducing metal oxides to their pure metal form.
Natural Occurrence of Hydrogen
Hydrogen primarily exists in the form of compounds and is not found in its elemental gaseous state in nature. For industrial applications, hydrogen must be produced through various processes, as it is not directly available in the atmosphere.
The emergence of the hydrogen economy introduces more sustainable processes into the chemical industry and plays a vital role in the generation of clean energy from H₂. Moreover, hydrogen is increasingly viewed as a foundation for sustainable development strategies, particularly in efforts to reduce CO₂ emissions and mitigate the greenhouse effect.
To better assess the environmental impact of hydrogen production, a color-coded classification system has been developed. This system categorizes hydrogen based on its production method, associated greenhouse gas emissions, and environmental sustainability, enabling consumers and businesses to make informed decisions about the hydrogen they use.
- 🔘 Grey Hydrogen: Produced through steam methane reforming (SMR) using fossil fuels—primarily natural gas—without capturing the resulting CO₂. This is currently the most common method of industrial hydrogen production, but it has a high carbon footprint.
- 🔵 Blue Hydrogen: Also produced using fossil fuels (typically SMR), but with carbon capture and storage (CCS) technology in place to trap CO₂ emissions. This method significantly reduces greenhouse gas emissions compared to grey hydrogen.
- 🟢 Green Hydrogen: Generated via water electrolysis powered by renewable energy sources (such as solar or wind). This is the cleanest form of hydrogen, with zero carbon emissions, making it essential for climate-neutral energy systems.
Industrial Applications of Hydrogen – The Core of Modern Production and Energy Transition
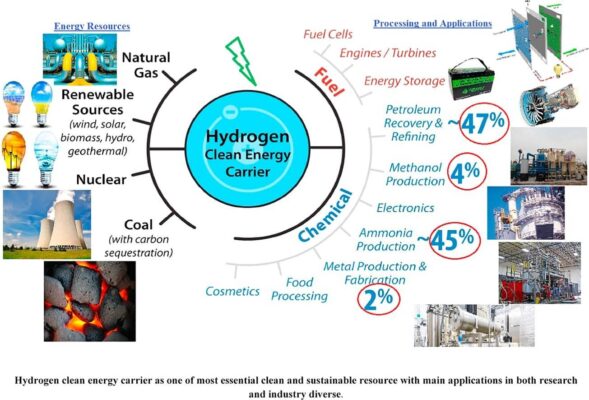
Refining & Petrochemical Industry
Hydrogen is a critical feedstock in the petrochemical sector, with refineries being the largest global consumers of H₂. It is used to process intermediate petroleum products and convert crude oil into cleaner, high-value fuels such as diesel, gasoline, and jet fuel.
Key Applications of Hydrogen in Refining:
- Hydrodesulfurization (HDS): Removing Sulfur Impurities
Stringent environmental regulations demand ultra-low sulfur content in transportation fuels. Sulfur compounds in fuel produce sulfur oxides (SOₓ) when burned, a major contributor to acid rain and photochemical smog, both of which severely affect air quality and public health. Hydrogen reacts with sulfur compounds to remove them as hydrogen sulfide (H₂S), thereby producing cleaner-burning fuels that meet modern emission standards.
- Hydroisomerization (HIP): Enhancing Fuel Quality, Hydrogen is used to convert straight-chain paraffins into branched-chain isomers, which improves fuel quality by increasing the octane rating in gasoline and the cetane number in diesel. This process boosts combustion efficiency and reduces engine knocking.
- Dearomatization: Reducing Aromatic Content, Hydrogen is used to saturate aromatic compounds (e.g., converting benzene to cyclohexane), resulting in less toxic, higher-value fuel components. This reduces harmful emissions and enhances fuel stability.
- Hydrocracking: Producing Lighter, Cleaner Fuels
In hydrocracking, hydrogen breaks down heavy hydrocarbons into lighter, more valuable fractions like diesel and naphtha. This process delivers higher yields, lower emissions, and cleaner fuels compared to conventional thermal cracking.
Through these hydrogen-based refining processes, the industry aims to improve fuel performance while significantly reducing harmful pollutants, supporting sustainable transportation and air quality goals worldwide.
Chemical Industry – Ammonia and Methanol Production
Ammonia Synthesis (Haber-Bosch Process)
Hydrogen is a key reactant in the industrial production of ammonia via the Haber-Bosch process, where nitrogen (N₂) from the air reacts with hydrogen (H₂) under high temperature and pressure in the presence of an iron catalyst:
N₂ + 3H₂ ⇌ 2NH₃ ΔH₀ = –45,9 kJ/mol
Ammonia (NH₃) is a vital precursor for the manufacture of fertilizers, pharmaceuticals, and plastics, supporting global agriculture and industry.
Methanol Synthesis from CO₂ and H₂:
CO₂ + 3H₂ ⇌ CH₃OH + H₂O ΔH⁰ = –49.8 kJ/mol
This pathway offers a carbon-neutral solution by recycling CO₂ emissions and converting them into valuable liquid fuel and feedstock, contributing to the circular carbon economy.
Metallurgical Industry – Green Steel Production
In the decarbonized steel industry, hydrogen serves as a clean reducing agent, replacing carbon (typically coke) in the direct reduction of iron ores. Key reactions include:
Fe₂O₃ + 3H₂ → 2Fe + 3H₂O
Fe₂O₃ + H₂ → 2FeO + H₂O
This hydrogen-based reduction emits only water vapor instead of CO₂, positioning hydrogen as the foundation for green steel production and low-carbon metallurgy.
Fuel Cells – Clean Energy from Hydrogen
Fuel cells generate electricity by combining hydrogen (H₂) and oxygen (O₂) in an electrochemical reaction that produces only water as a by-product:
This process converts chemical energy directly into electrical energy, offering high efficiency and zero carbon emissions at the point of use.
Key Applications of Hydrogen Fuel Cells:
-
- Fuel cell electric vehicles (FCEVs) — including cars, buses, trains, and even ships .
- Backup Power Systems: hospitals, data centers
- Renewable Energy Storage – Power-to-Gas (P2G)
The Hydrogen Economy – An Inevitable Development Trend
The Hydrogen Economy refers to a future energy ecosystem where hydrogen serves as a central energy carrier, replacing fossil fuels in a wide range of applications to enable a low-carbon and sustainable society.
Four Key Pillars of the Hydrogen Economy:
- Green Hydrogen Production
- Safe delivery and storage through pipelines, cryogenic tankers, or as hydrogen-based chemical carriers.
- Utilized in industrial processes, transportation systems, and power generation for clean, efficient energy conversion.
- Integration of carbon capture and utilization (CCU) to produce synthetic fuels and chemicals, enabling a circular carbon economy.Tái chế và tuần hoàn CO₂ thông qua phản ứng tổng hợp
VI NA Industrial Gases proudly supplies Air Products’ high-purity hydrogen, available in multiple grades to meet industrial and scientific standards: 99.995%; 99.9992%; 99.9995%; 99.9999%.
Contact us today for expert consultation on hydrogen products.

